Can silicon detect and fight cancer, turn a greenhouse gas like carbon dioxide into a fuel and form an inexpensive way to separate hydrogen from water? Is it possible to use the second-most abundant element in the Earth’s crust to create superior batteries for electric cars, low-cost lasers and literally dirt-cheap methods of detecting explosives?
Perhaps less important but no less cool, “Could we use silicon to make a contact lens glow?” asks synthetic chemist Jonathan Veinot, who leads the Veinot Research Group at the University of Alberta.
That may be a lot to ask from what starts out as sand on the beach. But as an expert in the synthesis, analysis and manipulation of silicon nanocrystals (also known as quantum dots) Veinot isn’t just posing the questions; he’s part of a Canadian-led renaissance in silicon research that is determined to come up with the answers.
To appreciate the magnitude of this work, a little context is in order.
We already know that silicon is the rock star of the electronics industry. The smart phones, computers, TV black boxes, game stations and other devices we love so much all exist because of the semiconductors manufactured from this plentiful metalloid. It also dominates the market for solar photovoltaics, which is held up as the great hope for our global transition away from fossil fuels. As one recent study co-authored by Veinot puts it, “It is an understatement that we live in a technological world made possible by silicon.”
While excellent at handling the flow of electrons, however, silicon has historically been a failed stage act when it comes to emitting light, unlike Group III-V and Group II-VI compounds like gallium arsenide and cadmium selenide, respectively. “Silicon is a different animal,” Veinot says. “Silicon itself shouldn’t be emitting light. It really shouldn’t.”
That’s because in bulk form silicon has an indirect band gap, meaning excited electrons and holes are more likely to emit heat than light when they recombine. For this reason, scientists just assumed silicon, the semiconductor miracle material, would never have optical or optoelectronic applications. When it came to light, it wasn’t even a dim star. It was a black hole.
Let There Be Light!
Then, British scientist Leigh Canham came along in the late 1980s. Canham, working at what was a government defence research group called the Royal Signals and Radar Establishment, used an electrochemical etching process to show that porous silicon could be made to emit bright light when an electric current was applied. “That created a shock around the world,” says nanochemistry pioneer Geoffrey Ozin, who leads his own team at the University of Toronto that is advancing the science of silicon quantum dots. Canham’s groundbreaking work, Ozin adds, influenced his own research early in his career. “Suddenly, we discovered semiconductor silicon can somehow break the rules.”
What followed, according to Ozin, was a frenzy of follow-on research as teams around the world tried to figure out why silicon could be made to do what was once thought impossible. Half a dozen theories emerged, but consensus formed around one: that silicon exhibits different chemical, physical and biological properties at the nanoscale. Just as important, silicon nanoparticles of varying sizes behave differently. Change their size, for example, and you can change their colour.
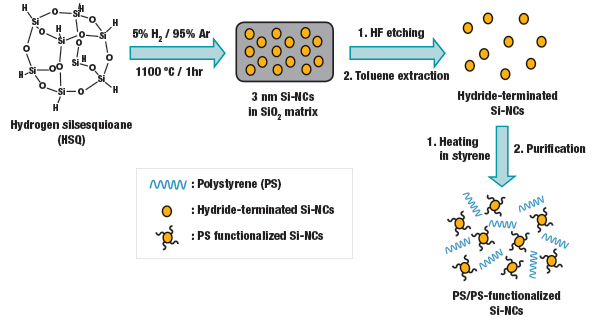
The Veinot Group synthesizes its silicon nanocrystals using thermal processing of hydrogen silsesquioxane (HSQ). Different processing temperature results in different sizes. In this case, an hour of processing at 1,100 C results in crystals that average three nanometres in size. Here, the nanocrystals are “functionalized” using polystyrene, resulting in a hybrid material. Photo Credit: Veinot Group
This was a big deal. Quantum dots made from Group III-V and Group II-VI compounds do the job well enough but the materials are expensive and the use of heavy metals mean they are toxic — in some cases highly so. Silicon, by contrast, is super cheap to produce and handle at high purities. Wafers cost just pennies to make and silicon is no stranger to existing microelectronics manufacturing processes, making it amenable to commercial-scale applications. Importantly, it’s also non-toxic. In fact, it was Canham who later discovered that porous silicon was biodegradable and generally benign to living cells, meaning in humans it is naturally broken down and excreted through urine. The possible applications for biomedical uses seemed endless.
Unfortunately, the excitement around silicon after Canham’s original discovery was short-lived. “People tried to make devices such as LEDs and lasers out of silicon and it never really came to anything. The field just sort of sputtered out,” Ozin says. Researchers turned their attention back to Group III-V and Group II-VI quantum dots, which both blossomed as areas of study.
Size Matters
What held back the potential of element number 14? Silicon nanocrystals (ncSi) produced by current methods are polydisperse, meaning that producing them results in particles of different sizes at the nanoscale — from one to 100 nanometres — with one to five nanometres showing the greatest quantum effects. This has forced researchers to measure properties based on an average of particle sizes, which is problematic, given the size-sensitive behaviour of these quantum dots. As a result, having polydispersed samples proved an obstacle to reliable experimentation, let alone the development of prototype products.

Veinot Group researchers are able to “tune” the size and surface chemistry of silicon quantum dots so that they emit different colours. In this image, nanoparticles suspended in bottles are tuned to display seven different colours. Photo Credit: Veinot Group
Think of silicon nanocrystals as balls used in sports. You have squash balls, golf balls, tennis balls, baseballs, footballs, soccer balls, basketballs and so on. They’re all balls but their size and unique surface makes them ideal for specific games. If you’re a baseball player practicing in a batting cage waiting for the next ball to be tossed, imagine your surprise if a basketball or golf ball gets launched your way. Needless to say, it will throw off your game.
This matters, particularly when someone’s life is on the line. Researchers such as Veinot have shown that silicon nanocrystals of a certain size and quality will heat up when exposed to light. Other research suggests that bioinert silicon quantum dots can be designed to target and penetrate cancer cells. Combine the two and you’ve got a potentially safe and powerful way to literally heat up and destroy dangerous tumours. But that’s only if all of the nanocrystals used are the right size as well as the same size.
On a similar note, we know that bulk silicon is generally considered harmless but at the nanoscale do we know for sure that particles don’t reveal toxic properties at specific sizes? The short answer is no, and the search for certainty hasn’t been an easy one. “There had been no convenient way to get tangible quantities, more than a gram, of silicon nanocrystals with well-defined size and shape,” says Veinot, who in the early 2000s was among a short list of researchers who emerged to tackle the challenge.
Road to Perfection
Toward the goal of getting monodisperse samples, Veinot’s chosen approach has relied on the thermal processing of hydrogen silsesquioxane (HSQ), which is easily produced in large amounts and already commercially available in the microchip industry. Veinot and his team found that a change in the reaction temperature controls the size of the resulting silicon crystals. The sweet spot for particles of three to 13 nanometres is between 1,100 C and 1,350 C, with particle size increasing as the temperature rises.
It’s not perfect — the resulting quantum dots can still be different sizes — but it significantly narrows the range to what Veinot describes as “near monodisperse.” Extending an earlier analogy, rather than get all the ball sizes in the sports world, you’re left with only ping pong and squash balls. To get even more size-specific, Veinot uses a scanning tunnelling microscope to zero in on and analyze the properties of individual particles, what U of T colleague Ozin calls a “clever” and “elegant” technique.
Such methods have significantly advanced the science. Still, developing fully reliable silicon nanocrystal-based applications requires the ability to produce — or isolate — monodisperse quantities. On this front, Ozin’s team raised the bar further, as outlined in “Small Silicon, Big
Opportunities: The Development and Future of Colloidally-Stable Monodisperse Silicon Nanocrystals,” published in 2012 in Advanced Materials.
Ozin’s group developed two techniques for separating monodisperse fractions of silicon nanocrystals from a polydisperse sample. One relies on a “density gradient” ultracentrifuge, which takes advantage of particle density. High spinning of the centrifuge tube causes denser larger particles to move near the bottom of the tube. Less dense smaller particles migrate to the top. Using this approach, Ozin’s team isolated fractions of nanoparticles from 1.3 to 2.2 nanometres in diameter. Another method, called size-selective precipitation, proved similarly effective.
Ozin says this new ability to produce monodisperse quantities brings the world a step closer to developing commercial application for silicon nanocrystals that can be labelled “green,” meaning the properties of particles used can be fully and accurately documented to assure they achieve their intended purpose without introducing unforeseen harm to human health or the environment.
The Great Manipulator
Size matters, of course, but in the world of silicon nanocrystals this is only part of the story. The optical properties of quantum dots can be influenced even further if they are functionalized by altering their surface chemistry. Which brings us back to Veinot. “There’s this surface dependence on the luminescence and that’s important because we can tailor the optical response,” he says. “We make it emit light and now we can make it emit any colour of light.”
Even small changes to the surface can bring about unexpected changes. Veinot recalls the time he slightly altered the surface of a silicon nanocrystal that had a red glow. He believed the change would quench the red light but instead the particle took on a pink glow. “Every time you turn a corner there are these surprises.”
In science, surprises are welcome for the new possibilities they bring. In one experiment, Veinot’s team figured out how to develop disposable, paper-based sensors to detect explosives, such as trinitrotoluene, or TNT. Filter paper was dipped into a concentrated solution of toluene filled with silicon nanocrystals with dodecyl-bonded surfaces. The researchers found that light emitted from the paper sensor was visibly quenched when exposed to nitroaromatics, even at nanogram levels.
Veinot’s group has also led development of silicon nanocrystal-polymer hybrid materials that can bring out the best properties of both materials, as well as new “synergistic interactions.” This could lead, for example, to new solar photovoltaic cell technologies that can convert a much wider range of the light spectrum into electricity, similar to high-cost, space-grade multi-junction cells made from layers of Group III-V compounds like indium gallium phosphide and indium gallium arsenide. Likewise, it could result in the development of new LED and laser technologies based on silicon nanocrystals. The group’s early work here has shown much promise.
The list of potential applications continues to expand as scientists get more focused with their investigations. In another example, it has been shown that silicon nanocrystals can split hydrogen gas from water without external stimulants such as light, heat or electricity, and that the smaller the nanoparticle the faster it works: for example, 10-nanometre particles proved 150 times better than those 100 nanometres in size. Based on these findings, we may one day see silicon quantum dots as part of an inexpensive way to boost the efficiency of hydrogen gas production.
Others are investigating the use of silicon nanoparticles to convert CO2 into useful chemicals and fuels, suggesting another pathway for recycling the world’s most notorious greenhouse gas. It has also been observed that silicon nanocrystals with a graphene shell can boost the charge capacity of lithium-ion batteries.
Some of these applications are being pursued as part of a Canadian-German collaboration spearheaded by Veinot. Called the Alberta/TUM International Graduate School for Hybrid Functional Materials, this NSERC-funded program brings together faculty and student researchers from the University of Alberta and Germany’s Technical University of Munich.
As well, Veinot is founder and president of Edmonton-based Applied Quantum Materials, a company he incorporated last year to commercialize some of the findings from his lab, with a heavy emphasis on biological imaging, for example, using silicon nanocrystals as a contrast agent for magnetic resonance imaging. Who knows, perhaps one day he’ll develop those glowing contact lenses?
Ozin, himself a major contributor to silicon nanocrystal science, doesn’t hesitate to give Veinot’s work high praise. He calls his surface chemistry work pioneering and refers to Veinot as one of Canada’s “shining” scientific lights.
“Chemists like John are invisible heroes. He’s basically unravelled all sorts of new chemistry and new physics about these materials that simply didn’t exist before,” Ozin says. “John is one of those people who is enabling nanosilicon so others can come along and develop the technologies.”
